Pfizer and BioNTech, the developers of the first effective coronavirus vaccine can prevent more than 90% of people from getting Covid-19 described the achievement as a “great day for science and humanity”.
The Covid-19 vaccine has already been tested on 43,500 people in six countries and no concerns have been raised. So the companies plan to apply for emergency approval to use the vaccine by the end of the month.
No vaccine has gone from the drawing board to being proven highly effective in such a short period of time. The announcement has been received with relief from the global community with the hope life could be back to normal soon.
More about the vaccine
Data shows that two doses, three weeks apart, are needed. The trials – in US, Germany, Brazil, Argentina, South Africa and Turkey – show 90% protection is achieved seven days after the second dose.
However, the data presented is not the final analysis as it is based on only the first 94 volunteers to develop Covid so the precise effectiveness of the vaccine may change when the full results are analysed.
Dr Albert Bourla, the chairman of Pfizer, said: “We are a significant step closer to providing people around the world with a much-needed breakthrough to help bring an end to this global health crisis.”
Prof Ugur Sahin, one of the founders of BioNTech, described the results as a “milestone”.
When will it be available?
The two companies say they will be able to supply 50 million doses by the end of this year and around 1.3 billion by the end of 2021. Each person needs two doses.
Pfizer and BioNTech say they will have enough safety data by the third week of November to take their vaccine to regulators.
Until it has been approved it will not be possible for countries to begin their vaccination campaigns.
Who gets the vaccine first?
Each country will decide how they prioritize it so not everyone will get the vaccine immediately.
It stands to reason that hospital staff and other medical workers will get it first as they are the most vulnerable, andthe elderly who are most at risk of severe disease.
It is not known yet if the vaccine stops you spreading the virus or just from developing symptoms. Or if it works equally well in high-risk elderly people.
Another question still to be answered is – how long does immunity last – this may take months or potentially years to answer.
There are also massive manufacturing and logistical challenges in immunising huge numbers of people, as the vaccine has to be kept in ultra-cold storage at below minus 80C.
The vaccine appears safe from the large trials so far but nothing, including paracetamol, is 100% safe.
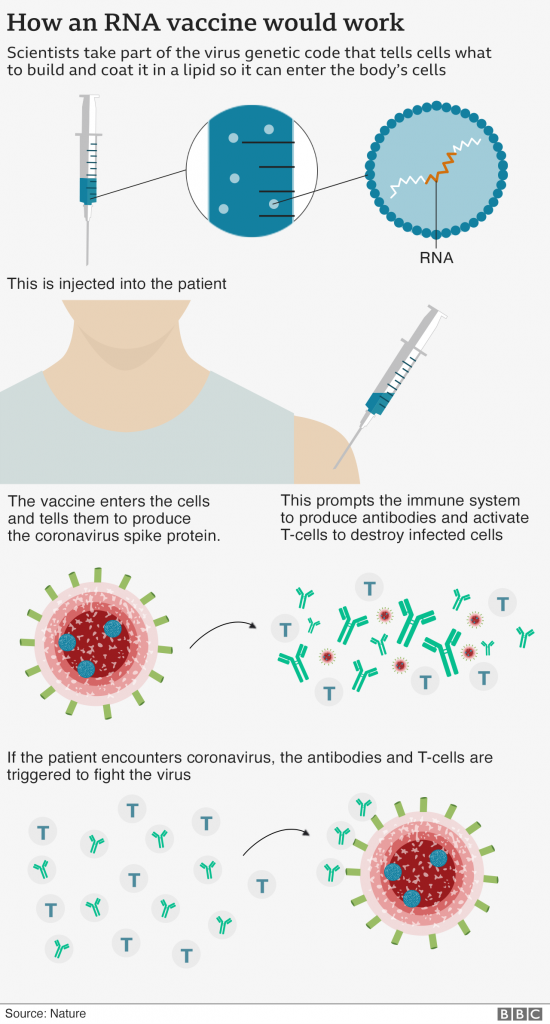
How does it work?
There are around a dozen vaccines in the final stages of testing – known as a phase 3 trial – but this is the first to show any results.
It uses a completely experimental approach – that involves injecting part of the virus’s genetic code – in order to train the immune system.
Previous trials have shown the vaccine trains the body to make both antibodies – and another part of the immune system called T-cells to fight the coronavirus.